Description
Phosphate Buffered Saline (Non-Biologix)
Research-Grade Buffer Solution (PBS)
Tagline: Standard Laboratory Buffer for Biological Research
Product Description
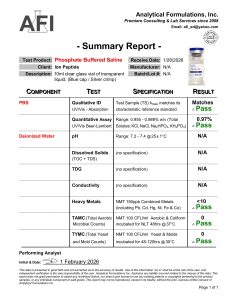
Phosphate Buffered Saline (PBS) is a balanced salt buffer solution widely used in biological and biochemical research laboratories. PBS is an aqueous isotonic solution of phosphate and saline, designed to maintain a stable pH and osmotic balance similar to physiological conditions. It contains salts such as sodium chloride, sodium phosphate, and potassium phosphate, providing a gentle environment compatible with many experimental systems.
This buffer solution is commonly used in cell culture workflows, molecular biology protocols, and general laboratory procedures where maintaining a neutral pH and isotonic environment is critical.
For Laboratory and Scientific Research Use Only. Not for Human Consumption.
Key Features
-
Stable physiological pH (~7.2–7.4)
-
Isotonic salt composition similar to biological fluids
-
Non-toxic to cells in short-term applications
-
Water-soluble and easy to use
-
Suitable for a range of biological assays
-
Available as ready-to-use liquid or concentrate
Important Note
For laboratory and scientific research only. Not for human consumption, diagnostic, or therapeutic use.