Description
Bacteriostatic Water (0.9% Benzyl Alcohol)
Research-Grade Diluent
Tagline: Sterile Solution for Laboratory Reconstitution
Product Description
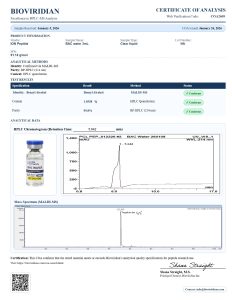
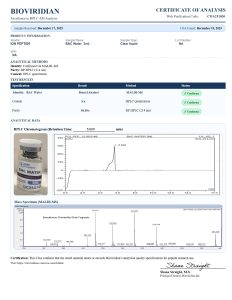
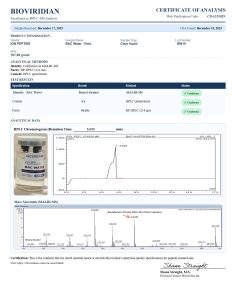
Bacteriostatic Water is a sterile, non-pyrogenic solution of water with 0.9% benzyl alcohol as a bacteriostatic preservative. It is widely used in laboratory research for diluting and reconstituting lyophilized compounds before in vitro or in vivo use.
Researchers choose bacteriostatic water peptide to enable multiple withdrawals from a single vial while maintaining sterility, making it ideal for controlled research environments.
For Laboratory and Scientific Research Use Only. Not for Human Consumption.
Why Researchers Choose Bacteriostatic Water Peptide
-
Multi-Use Vial: Benzyl alcohol preservative allows repeated withdrawals under aseptic conditions.
-
Sterile & Non-Pyrogenic: Meets USP standards
-
Versatile: Suitable for reconstituting peptides, proteins, and other lyophilized reagents.
-
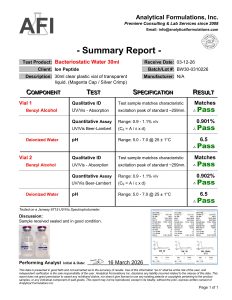
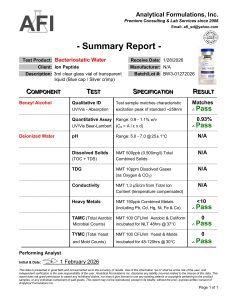
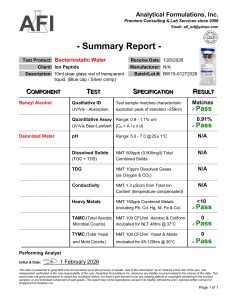
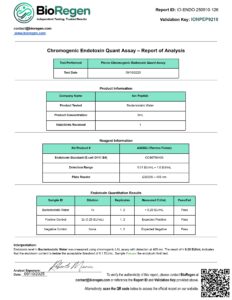
Batch Verified: Each lot tested for sterility, clarity, and preservative content.
Important Note
For laboratory and scientific research only. Not for human consumption, injection, or clinical use.